 |
|
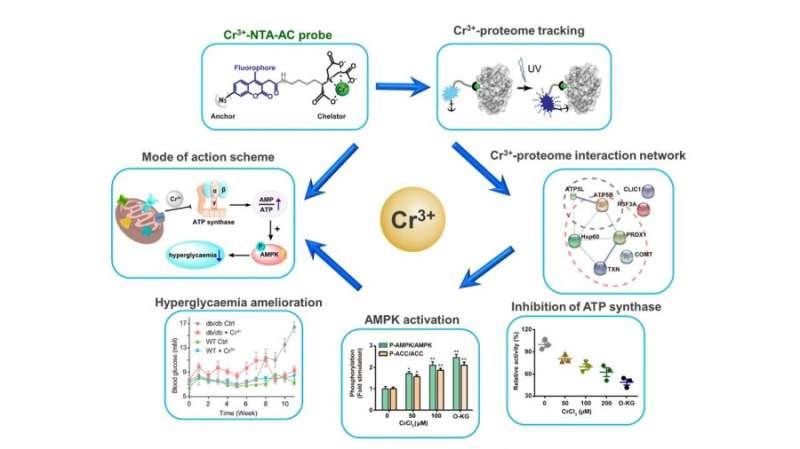
Figure 1. Chromium(Cr3+) fluorescent probes can label Cr(III)-binding proteins specifically in cells (top), Cr(III)-binding proteins are concentrated in the regulation of mitochondrial biological functions (right), and Cr(III) reduces ATP synthase Enzyme activity (bottom right), Cr(III) initiates downstream AMPK pathway (bottom), Cr(III) improves glucose metabolism (bottom left), diagram of Cr(III) mode of action (left). |
A research team led by Professor Hongzhe Sun from the Department of Chemistry, The University of Hong Kong, has published a paper titled - Mitochondrial ATP synthase as a direct molecular target of chromium(III) to ameliorate hyperglycaemia stress in Nature Communications.
For more than half a century, the importance, pharmacological properties, and mechanisms of action of Cr(III) in human physiology have been debated. The originally conceived low-molecular-weight chromium(III)-binding peptide (chromodulin) has never been reproducibly identified. Cr(III) is widely believed to improve glucose metabolism, maintain normal blood sugar levels, regulate carbohydrate and lipid metabolism, and enhance insulin signaling. To uncover the protein targets of Cr(III) and elucidate the molecular mechanism, the team developed a fluorescent probe for detecting transient metal-protein interactions, achieving a high spatiotemporal resolution tracking of the Cr(III) proteome in HepG2 cells.
The researchers discovered that Cr(III) binds residues of Thr213 and Glu242 in ATP synthase's active site, replacing magnesium ion (Mg2+), reducing ATP synthase activity, and leading to AMPK activation. The AMPK pathway then alleviates mitochondrial damage caused by high glucose and improves glucose metabolism. This mode of action was successfully verified in a mouse model of type II diabetes.
The team then revealed that Cr(III) replaces magnesium ions (Mg2+) in ATP synthase, reduces ATP synthase activity, and activates the downstream AMPK pathway, resulting in improved glucose metabolism. Building on their previous work, the team synthesized a fluorescent probe for tracking and recognizing Cr(III)-binding proteins in living cells.
This study addresses how Cr(III) improves hyperglycemic stress at the molecular level for the first time. With the identification of multiple Cr(III)-binding proteins, this study also opens a new horizon for further investigation of the pharmacological role of Cr(III) in other diseases other than diabetes and opens new horizons in tackling anti-neurodegenerative diseases and anti-aging.
Link of journal paper can be accessed from: https://www.nature.com/articles/s41467-023-37351-w
